Venous Flow Through Flaps
Anatomic considerations
Authored by Darrell Brooks, MD and Rudy Buntic, MD
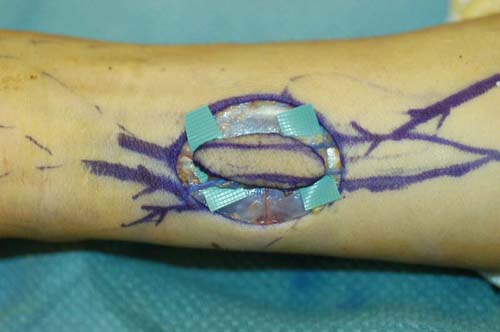
Soft tissue coverage of the hand presents an ongoing challenge to the reconstructive surgeon. Skin grafts are not always appropriate, local flaps are not always available, and distant pedicle flaps are often too morbid. When conservative approaches are not available or have failed, microvascular transplant of an arterialized venous "flow-through" flap (VFTF) provides a unique and creative option for difficult reconstructions of the hand.
Musculocutaneous flaps are appropriate for covering larger soft tissue defects but can be bulky and restrictive when applied to the wrist, hand, and digits. Fasciocutaneous flaps are thin and pliable. However, their transplant not only requires sacrifice of a peripheral artery, but can lead to significant vessel mismatch when anastomosed to distal vessels. In general, selection of the best flap for a particular reconstruction balances optimizing form and function at the recipient site with minimizing the morbidity at the donor site.
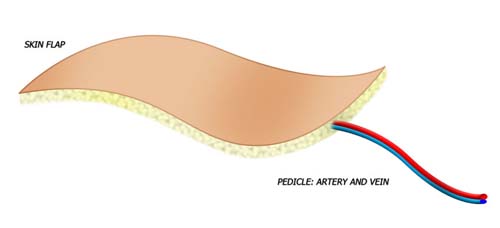
Venous flow through flaps are thin and pliable with vessels similar in size to those of the hand. Their harvest results in minimal donor site morbidity. These flaps are composed of skin, subcutaneous tissue, and a plexus of veins. Unlike conventional flaps where the nutrient capillary beds are supplied by an inflow artery and drained by an outflow vein, the VFTF has no native arterial inflow circuit. All flow proceeds into and out of the flap via the venous plexus.
The exact physiology of VFTF survival has not been determined. Three theories have been proposed:
- Reverse Shunting
- Reverse Flow
- Capillary Bypass
In the conventional flap, nutrient capillary beds are supplied by an inflow artery and are drained by an outflow vein. The conventional flap tends to be thicker, and is usually well defined in terms of both inflow and outflow by a named artery and its venae commitans.
In the venous flap, a vein delivers inflow of blood and outflow of blood. The flap tends to be thinner, can be designed over numerous veins, and often does not have a specific anatomically named inflow and outflow vein.
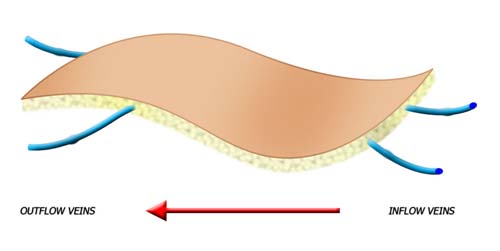
Venous flap perfusion is derived from inflow via veins, rather than arteries, and outflow via veins.
Anatomy and Physiology
The physiology of the VFTF places unique restrictions on its design. The tissue furthest away from the venous plexus is prone to congestion and necrosis. Larger flaps require a more extensive venous plexus for complete survival. Studies have shown that VFTFs designed with a central venous plexus with two or more efferent veins have a survival pattern similar to that of a conventional flaps which have an inflow artery and an outflow vein.
A fine network of veins extending throughout the flap is not the only factor in the VFTF's survival. Characteristics of the donor site also play a role. There are several potential VFTF donor sites:
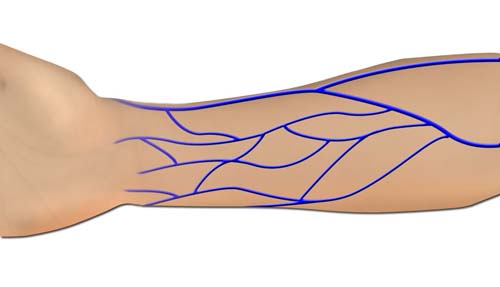
- Distal Volar Forearm
- Proximal Volar Forearm
- Dorsum Digit/Hand
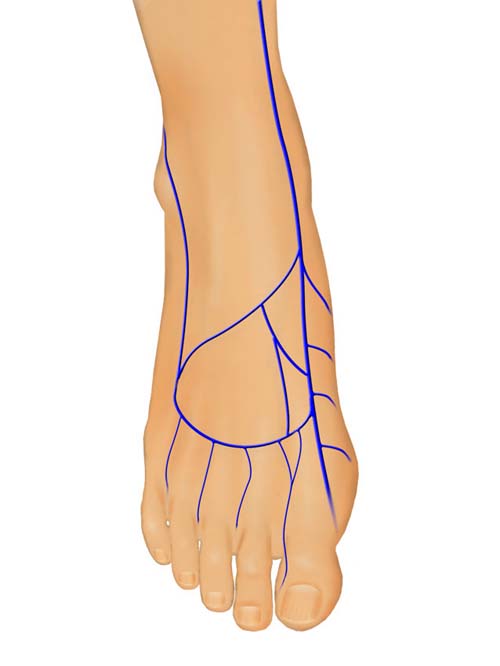
- Dorsal Foot
- Medial Thigh/Leg
- Upper Arm
The superficial venous system located distally on the extremity is less likely to have valves, has more extensive networking, and is more intimately associated with and supportive of its overlying skin. This improves the success rate and makes the hand, foot, and distal volar forearm preferred donor sites for VFTFs. When a larger flap is required the proximal forearm is the next best option. Direct visualization of the venous plexus through the thin skin of the distal extremities allows precise design of the VFTFs. The flap can not only be centered over the most appropriate plexus, but creative inflow and outflow circuits can also be designed in the branching venous system. The donor sites of small and moderate sized flaps can usually be closed primarily.
VFTFs harvested from the leg and upper arm are nourished by the saphenous and basilic vein respectively. These flaps are useful when long vascular conduits or a larger soft tissue paddle is required. These flaps are associated with increased subcutaneous tissue between the nourishing vein and overlying skin. The smaller venous systems cannot be visualized and their extent cannot be determined at the time of flap design. These flaps are usually designed over the main vein. Their maximum width is restricted to insure optimal survival.
Another limiting factor in the survival and success of VFTFs is the recipient bed. Areas with ongoing infection can prolong healing time and delay neovascularization of the VFTF. This can prolong the time the flap has to rely on venous physiology and potentially decrease flap survival. In addition, factors such as infection can result in activation of platelets and increase thrombosis potential.
The VFTF can be harvested with additional tissues creating a composite VFTF. This composite flap is more difficult to design because success is dependent on spatial distribution of multiple components rather than just inflow and outflow circuits. Sensory nerves such as the brachial cutaneous and saphenous nerves have been included to create a sensate flap or to graft nerve defects at the recipient site. Tendon has been included to reconstruct tendons, ligaments, and joints capsules. One author has reported inclusion of tibial bone with a saphenous venous flap for reconstruction of soft tissue and bony defects in the hand. Theoretically, these are vascular grafts. Composite VFTFs have been reported sporadically but no series has evaluated the efficacy of their components as vascularized grafts.